A tribofilm refers to a thin layer that forms in a mechanical application due to interaction between lubricant molecules and the lubricated surfaces. This definition is flexible but typically refers to films on surfaces that form because of ionic interaction or adsorption. It can also be thought of as a “third material” that is neither the lubricant nor the machine surface, but rather an intermediate layer formed by the interaction of both.
This concept is similar to others in surface interactions – for example in aerodynamics we often talk of a boundary layer of air that separates the regular flow stream from the aircraft surface.
Different types of tribofilms
There are two major types of tribofilms:
- Tribochemical reaction films
- Polymeric and non-sacrificial reaction films
Reaction films are generally those created by the reaction of antiwear or EP-style additives with metals surfaces.
Polymeric and non-sacrificial reaction films are those similar to friction modifiers or the weak binding between an ester and a surface.
Formation of reaction-type tribofilms
The reactive additives in a lubricant (EP or antiwear) initially adhede to the surface. The surface will attract the polar components (sulfur, oxygen) and under pressure, the bonds between zinc and sulphur break. The result is a reaction of the sulfur and the phosphorous components with metal surfaces.
This creates a glass polyphosphate over time. This anti-wear film is very small, around 50 to 150 nanometers.
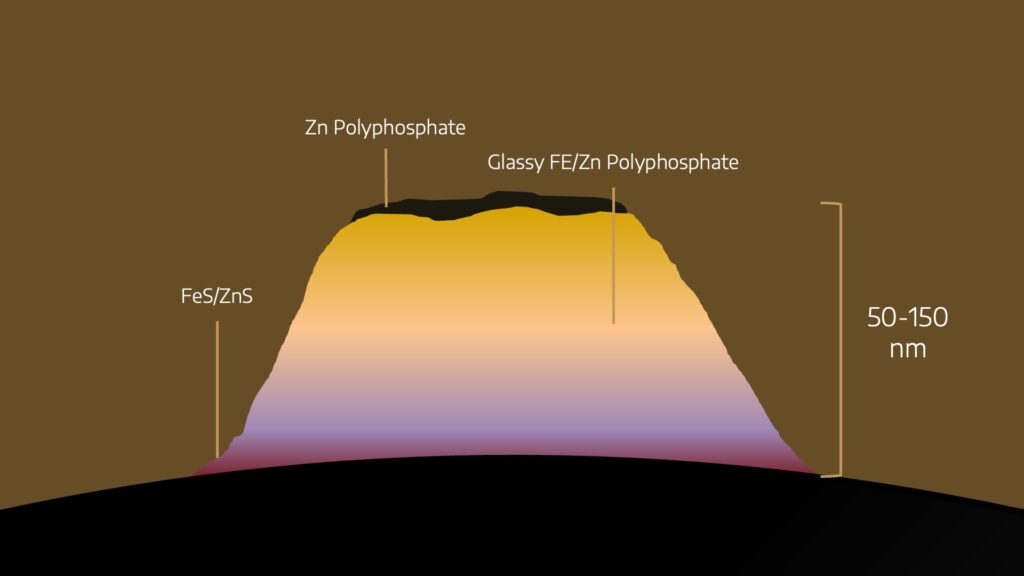
These sacrificial films therefore form a hard barrier above the component surface, protecting it from abrasive / adhesive wear in highly loaded application.
Formation of polymeric-type tribofilms
Ester base oils are a good example of molecules that form polymeric-type tribofilms. The polarity of the oxygens in the ester functional group helps adhede the molecules to the equipment surface. While the adheded molecules do not form solid or semi-solid materials in the same way as antiwear films, the bond strength is sufficient enough to create a “boundary layer” that exists between the bulk oil and the surface.

