Gas compressors are used to compress many different gases and mixtures. These range from inert gases like hydrogen, nitrogen, or helium, to reactive gases such ammonia, chloride, or hydrogen sulphide. Among the most common are air and hydrocarbon gases like methane or propane. Gas mixtures can contain moisture or acid components like hydrogen chloride or heptane. The viscosity selection for a lubricant is the most important property of the compressor oil (as base oil viscosity determines the lubricating regime at the bearings and sliding contacts) but the solubility of process gases in oil can have a significant impact on oil viscosity.
If the physics and chemistry of the gas-lubricant interaction is not managed correctly, the result can include damaged bearings or rotors, short lubricant life, varnish and deposit formation, form formation, downstream catalyst damage and increased oil consumption. This can often lead to a lengthy and expensive compressor downtime, and subsequently, standstill of the entire plant.
This main impacts of the interaction between gas and compressor oils are:
- Significant viscosity alteration as the gas dissolves into the oil
- Reactions between the lubricant and reactive gases that can cause decomposition
- Oil consumption as the oil is carried through the process stream
VISCOSITY ALTERATION
When gases dissolve into liquids they can radically alter the bulk viscosity of the fluid. This is reasonably intuitive, given a comparison of viscosities:
- Absolute viscosity of methane at 40oC = 0.0116 cP
- Absolute Viscosity of ISO 460 mineral oil at 40oC = 396 cP
The fact that there are 4 orders of magnitude difference suggests that any mixture would have a substantially lower viscosity than the neat oil; this is borne out by experimental and field data, in which absolute viscosity reductions of almost 50% have been observed.
Prediction of gas solubility in lubricants is a difficult exercise, and typically relies on the end-user asking the lubricant manufacturer for experimental data measured for a particular lubricant in use. However, there are some general principles. Just as “like dissolves like” means that polar liquids tend to dissolve in polar, so too do polar gases dissolve in polar liquids.
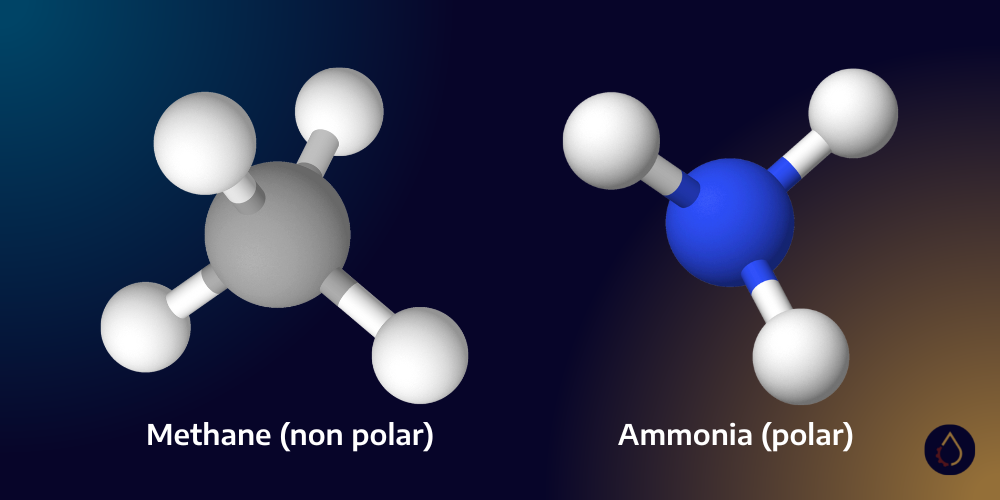
Methane is an excellent example; it is a non-polar hydrocarbon molecule that will dissolve into other non-polar hydrocarbon molecules. This generally excludes the use of standard mineral oils from use in natural gas compression due the effect of gas dilution. Methane can be dissolved at equilibrium at high temperatures and in high viscosity oils for a prolonged period. This is why you will see large Ariel reciprocating compressors being lubricated by polyalkylene glycols. Being a polar liquid, methane is not dissolved (as much) in PAGs.
Methane can still enter the bulk fluid during the compression cycle however, it will remain as an isolated bubble until it eventually percolates out of solution
While user of PAGs in the natural gas industry may be unhappy about the maintenance cost and the extra cost of PAG fluids, they are extremely stable for gas compression applications.
The reverse is also true. Ammonia is a common refrigerant and is a polar molecule that will readily dissolve in water (also a polar liquid). PAGs would represent a terrible choice for Ammonia compression because the gas readily dissolves in the bulk oil, reducing its viscosity. It’s for this reason that PAO synthetic oils are a common choice in Ammonia compression.
There is another reason that PAGs would make a poor choice for the compression of ammonia, and that is its decomposition characteristics.
DECOMPOSITION REACTIONS
Compressors are a unique lubricating application because of the variation in gases that are in the process stream. The most common gas, air, is of course relatively inert. But as we move to more reactive gases like pure oxygen and chlorine they chemically interact with the lubricant molecules – sometimes with very negative consequences.
Esters are no longer a viable option. Nor are PAGs, which hydrolyze with strong bases and highly reactive chemicals. Alkylated Naphthalenes will eventually undergo an aromatic substitution reaction. Mineral oils are off the table because any olefins (unsaturated site, or in more simple terms, double bonds between carbons) will be oxidized by the reactive species. It might be tempting to opt for a hydrotreated / hydrocracked base oil package that has a high degree of saturation, but in service mineral oils tend to generate olefins as a decomposition product. The result is a coupling reaction; once started the molecular weight and viscosity start to rise.
Adverse reactions are not limited to the reactive gases either. Whilst ammonia is not “reactive” in the same manner as chlorine, it can act as a Lewis base – reacting with carboxylic acids at high temperatures to produce amides. They can also undergo acid-base neutralization reactions with acidic additives in the oil (typically the corrosion inhibitors) or react with oxygen at high temperatures to form NOX compounds that act as oxidation catalysts.
This is the challenge of lubrication technologies – it is chemistry meets mechanical engineering meets fluid dynamics.
OIL CONSUMPTION
Strangely, the oil can be responsible for high oil consumption. It is frequently the role of the compressor oil to not only lubricate but also to remove some of the heat generated by the compression process and cool the compressor’s gas flow. Temperatures in excess of 90oC are common in many compressor applications and this exceeds the boiling point for many light ends in lubricant formulations.
Any vapours that are formed are subsequently transported downstream along with the gas flow. Unlike oil droplets, gaseous phases are not captured by oil separators, and this volume does not return to the lube system. So at high temperatures (or alternatively, at high compression rates) the volatility of the oil becomes an important determining factor to oil consumption.
Just as solubility rules explain significant changes in oil viscosity, oil absorption into the gas stream is governed by the same principles. The compressed gas may absorb oil molecules which become entrained in the gas flow. The significance to plant asset management is that many chemical processes require downstream catalysts to further initiate reactions that transform the process gas. If the process gas now contains some component of lubricating oils these will reduce or even eliminate the effectiveness of these catalysts by “poisoning” them. Catalyst replacement is frequently a costly and time-consuming exercise, so any reduction in catalyst life can have a major impact on overall plant profitability metrics.
KEY TAKEAWAYS
The interactions between the gas being compressed and the lubricating oil are sometimes difficult to predict. It can require specialist knowledge of chemical reactions, as well as experimental data available only to the lubricant manufacturers. Great care should be taken when selecting an appropriate compressor oil for the application – when in doubt, consult a specialist.
